Big Claims, Thin Evidence: What the Latest Medical Cannabis Study Really Shows
A major cannabis review has sparked headlines, but the evidence is more nuanced. Mamedica explores what the latest study says, where the gaps remain, and why real-world data matters.
A major new review has sparked fresh headlines around medical cannabis and mental health, with some reports presenting the findings as a clear-cut verdict. The reality is more complicated. While the paper raises important questions about the strength of current research, it also exposes just how narrow and incomplete much of the evidence base still is.
Published in The Lancet Psychiatry, the review analysed 54 randomised controlled trials involving 2,477 participants. It found no significant clinical benefit for anxiety disorders, PTSD, psychotic disorders, OCD, anorexia nervosa or opioid use disorder, and noted there are still no randomised controlled trials assessing cannabinoids as a treatment for depression. Most outcomes were rated low or very low certainty, while nearly half of the included studies carried a high risk of bias.
Limited evidence is not the same as no value
One of the biggest problems in this space is how quickly limited evidence is translated into no clinical value. Those are not interchangeable ideas. Low-certainty evidence means confidence in the findings is restricted. It signals an evidence base still in development, not one that has delivered a final answer.
This distinction is especially important in mental health and chronic illness. Patients rarely present with one neatly defined condition. Anxiety can sit alongside poor sleep. Chronic pain can affect mood, concentration and resilience. Depression can overlap with inflammation, fatigue and long-term treatment resistance. Clinical reality is often far messier than the tightly controlled settings used in randomised trials.
What the latest review leaves out
One of the clearest gaps in the current evidence is depression. Despite how common it is, the review found no randomised controlled trials assessing cannabinoids as a treatment for depression. That absence alone should encourage more caution in how the findings are interpreted publicly.
Scope is another issue. Much of the discussion around the paper centres on psychiatric conditions, yet one of the biggest real-world reasons patients explore medical cannabis is chronic pain. Pain rarely exists in isolation. It often appears alongside sleep disruption, anxiety, low mood and reduced quality of life. When studies isolate one diagnosis and exclude wider complexity, they can miss how people actually experience illness.
The gap between research design and patient reality
Randomised controlled trials remain vital, but they are built to reduce variability. They usually focus on narrowly defined patient groups, fixed doses and short timeframes. Scientifically, that has real value. Clinically, it can limit relevance when the goal is to understand how treatment works in everyday practice.
In real-world settings, patients are often managing multiple treatment-resistant conditions at once. Symptoms change over time. Treatment pathways are adjusted. Outcomes do not always follow a neat or linear pattern. This is where the current conversation around evidence often falls short. The people entering clinics are more complex than the populations many studies are designed to capture.
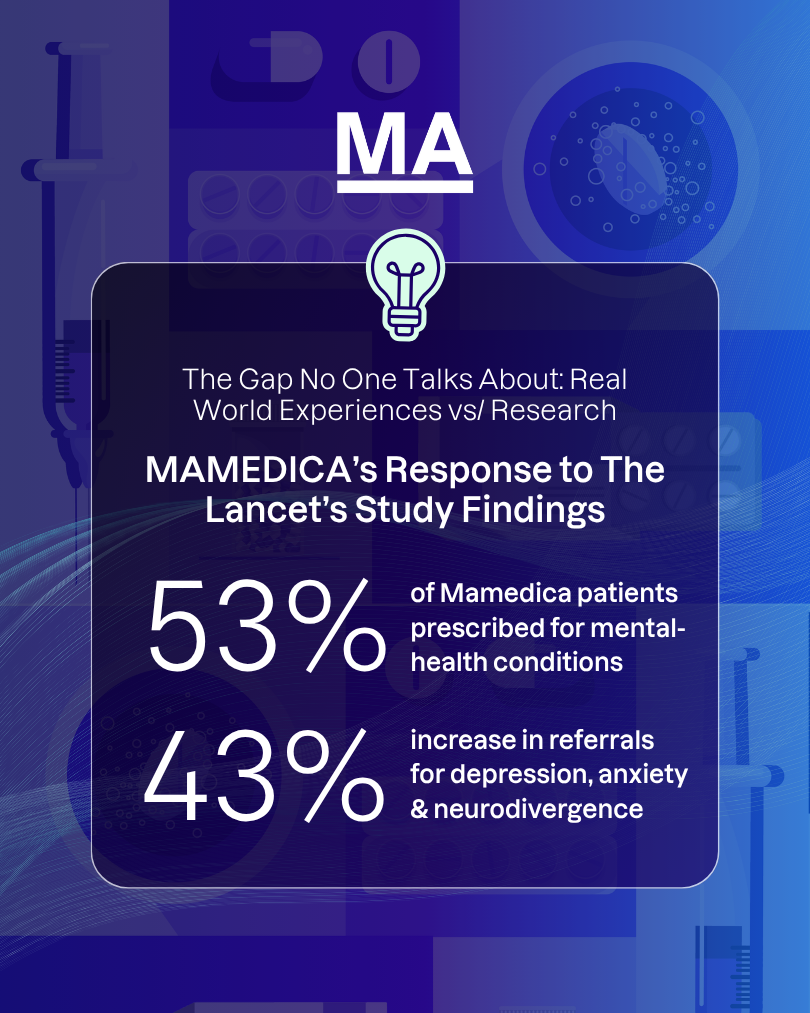
What Mamedica is seeing in practice
This disconnect is not theoretical. It is visible in referral patterns and prescribing data. At Mamedica, 53% of patients are prescribed for mental health conditions, while referrals for depression, anxiety and neurodivergence have risen by 43%.
These figures add another layer to the latest study headlines. They show a level of demand that extends beyond narrow trial categories and reflects the more complex symptom picture seen in practice. Many patients are not seeking support for one isolated issue. They are living with overlapping challenges affecting mood, sleep, focus, pain and day-to-day function.
This is exactly where real-world evidence becomes more valuable. When patient need is evolving faster than the research base, there is a stronger case for studies that mirror how people actually present in clinic rather than how they are filtered for controlled trials.
Chronic pain remains one of the biggest drivers of demand
For many patients, chronic pain is the starting point. It affects mobility, sleep, employment and mental wellbeing, particularly through mid-life and beyond. At Mamedica, more than 40% of patients seek support for chronic pain, usually after exhausting conventional treatment pathways.
This sits within a broader picture of chronic illness affecting working life across the UK. Mamedica’s Health & Work Index found that one in five UK adults have left a job due to illness. The figure rises to 24% among women and 37% among lower-income groups. A further 18% have reduced their working hours because of an ongoing health condition. Among people living with long-term illness, 23% say they have felt pressured to return to work before they were well enough.
These are not marginal quality-of-life concerns. They speak to the wider effect chronic conditions can have on income, independence and recovery.
The wider evidence base is still mixed
The latest review is not the only important paper in this area, and the wider evidence remains mixed. Some systematic reviews suggest modest benefits for certain symptoms or conditions, while also pointing to low-certainty findings and relatively common side effects such as dizziness, fatigue and drowsiness.
This does not support blanket claims of effectiveness across every indication. It also does not justify an overly simplistic dismissal of cannabis-based medicines altogether. The more accurate reading is that this remains a developing field, with outcomes shaped by indication, study design, dosing, patient selection and duration of follow-up.
For clinicians, policymakers and patients alike, the challenge lies in interpreting uncertainty without overstating it in either direction.
Why better research now matters
The central issue is not whether scrutiny is needed. It is whether the research framework can catch up with the reality of patient need. Better evidence means more than simply adding more trials. It means larger, more representative studies, longer follow-up periods, clearer reporting on dose and formulation, and stronger inclusion of patients living with overlapping, treatment-resistant conditions.
Real-world evidence has an important role here. It should not replace randomised controlled trials, but it can help build a fuller picture where trial evidence remains narrow, missing or poorly aligned with clinical practice.
With demand continuing to rise, the gap between controlled evidence and lived patient reality is becoming harder to ignore.
Looking beyond the headlines
The boldest headlines often suggest certainty where little exists. The latest review raises valid concerns around research quality and overstatement, but it also highlights a deeper problem: current evidence still does not fully reflect the complexity of patient need.
For people living with chronic pain, insomnia, anxiety, depression and other long-term conditions, the real question is not whether careful scrutiny is warranted. It is whether research, policy and treatment pathways can evolve quickly enough to reflect real-world care.
Right now, that work is still underway.










